The Periodic Table - Reading It
Using The Periodic Table
The Periodic Table of Chemical Elements arranges all of the known chemical elements in an informative array. Elements are arranged from left to right and top to bottom in order of increasing atomic number. Order generally coincides with increasing atomic mass.
The rows are called periods. The period number of an element signifies the highest energy level an electron in that element occupies (in the unexcited state), according to the Los Alamos National Laboratory. The number of electrons in a period increases as one moves down the periodic table; therefore, as the energy level of the atom increases, the number of energy sub-levels per energy level increases.
Elements that occupy the same column on the periodic table (called a "group") have identical valance electron configurations and consequently behave in a similar fashion chemically.
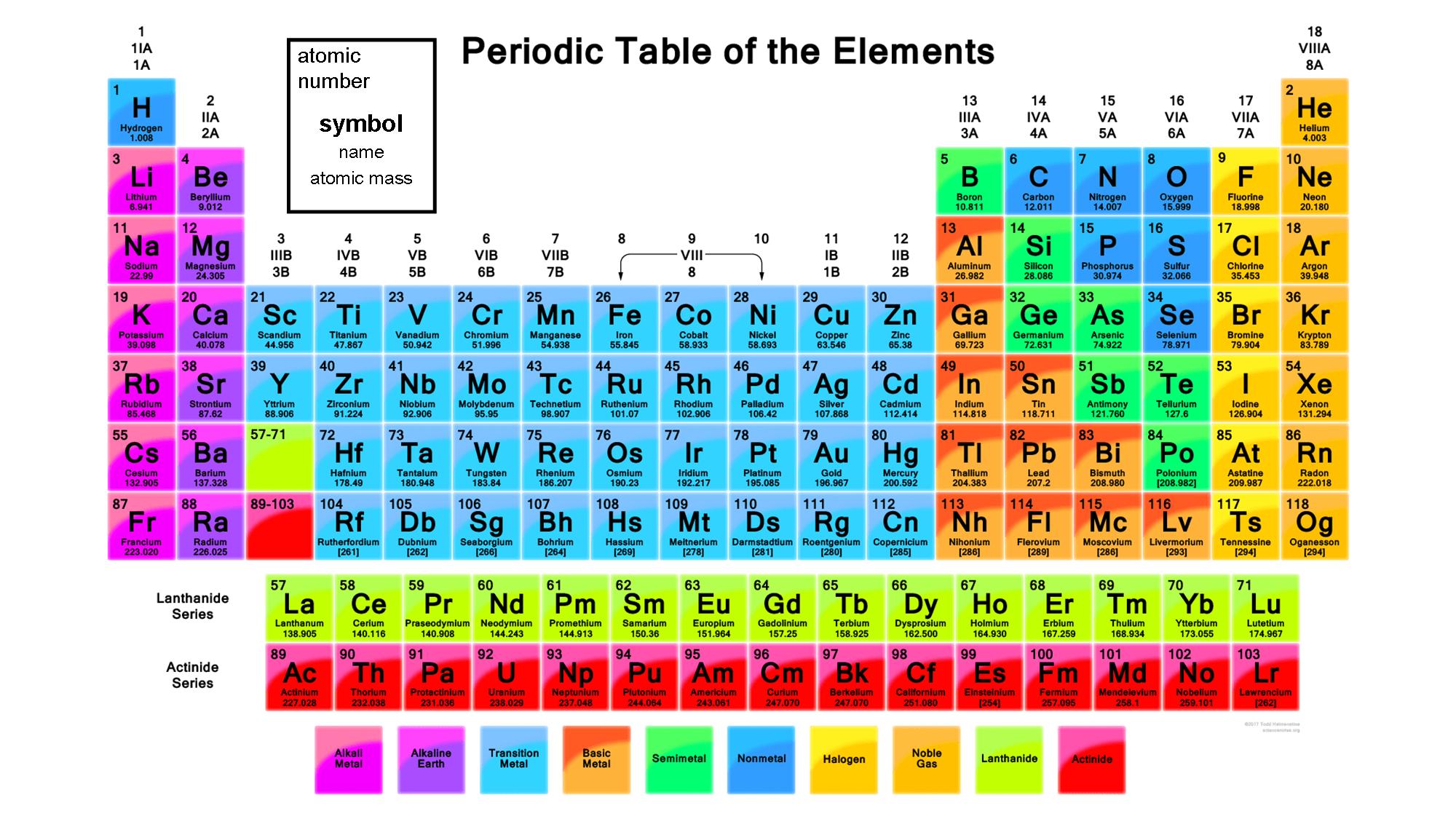
For each element, the Periodic Table indicates very important informations:
Atomic symbol - The atomic symbol (or element symbol) is an abbreviation chosen to represent an element ("C" for carbon, "H" for hydrogen and "O" for oxygen, etc.). These symbols are used internationally and are sometimes unexpected. For example, the symbol for tungsten is "W" because another name for that element is wolfram. Also, the atomic symbol for gold if "Au" because the word for gold in Latin is aurum.
Atomic number - The number of protons in an atom is referred to as the atomic number of that element. The number of protons defines what element it is and also determines the chemical behavior of the element. For example, carbon atoms have six protons, hydrogen atoms have one, and oxygen atoms have eight.
Atomic mass - For naturally occurring elements, the standard atomic mass of an element is the average mass of the element in atomic mass units (amu). Individual atoms always have an integer number of atomic mass units; however, the atomic mass on the periodic table is stated as a decimal number because it is an average of the various isotopes of an element. The average number of neutrons for an element can be found by subtracting the number of protons (atomic number) from the atomic mass. For lab-created trans-uranium elements — elements with atomic numbers higher than 92 — there is no "natural" abundance. The convention is to list the atomic mass of the longest-lived isotope in the periodic table. These atomic weights should be considered provisional since a new isotope with a longer half-life could be produced in the future.
Back to The Periodic Table